Project Results
A course-based undergraduate research experience (CURE) is described, both in its original, field-based and in-person design, as well as its adaptation to online delivery during the COVID-19 pandemic. The CURE experience was centered around the topic of microbes attaching to ocean plastic debris, and included connecting nontraditional undergraduate students attending a primarily teaching institution to personnel of a research-intensive institution. The CURE was implemented in nonmajors and majors’ general biology laboratory courses, as well as in an introductory microbiology lab course for health majors. Student perception of the CURE was assessed quantitatively through self-administered retrospective surveys, and qualitatively using open-ended survey questions and focus group interviews. Survey questions were grouped into four themes: (1) research at the research institution, (2) research at the teaching institution, (3) laboratory skills, and (4) interest in science. To assess impact of the CURE by modality, major and course level, theme scores were analyzed using ANOVAs (α = 0.05). Changes in student perception of learning were measured by comparing the “before” and “after” survey scores in each theme. The main source of variation in the model was time, followed by theme and major, while modality had a negligible effect. Overall, there were increases in student perception of learning across all themes across modality, major, and course level; however, not all were statistically significant. Highest gains were observed among nonmajors, while those of the majors’ upper division courses were the lowest. On the other hand, majors’ overall scores were the highest. Health majors’ survey gains were mostly between those of nonmajors and majors. Qualitative data contextualized the quantitative results. The online adaptation was a valuable alternative, especially for nonmajors, as it expanded the range of students reached, with results similar to the in-person alternative. Preliminary data show a positive effect of the CURE in student success majoring in biology.
About This Project
While many studies have focused on the visible impact of ocean plastic, we know much less about the impact of microbes that choose plastic as their home, forming the so-called plastisphere. The goal of my project is to evaluate the microbial diversity on floating plastic such as retail plastic bags in coastal waters and its potential impact on humans living on coastal areas. The project uses metagenomic next generation sequencing and standard microbiology techniques.
Ask the Scientists
Join The DiscussionWhat is the context of this research?
A recent report estimates the amount of plastic in the ocean to be on the order of 150 million tons, with production doubling over the next 20 years. Worldwide, nearly two million single-use plastic bags are used each minute. Plastic affects marine organisms big and small, and the existence of unique microbial populations on minuscule plastic debris called microplastics has been described. However, little is known about the nature of microbes attaching to plastic in beach water. My research focuses on studying microbes populating floating plastic litter that we often see around beaches. Enrichment in potentially pathogenic genera such as Vibrios, could be a health risk for coastal residents and visitors. We could also find microbes degrading plastic!
What is the significance of this project?
To our knowledge no similar studies exist. There are studies on microbial diversity on immersed surfaces of various materials, mainly from a biofouling studies, as well as on microplastics floating in ocean garbage patches. However, no metagenomic studies exist on microbial populations attaching and living on floating plastic material. The methods used are current (next generation DNA sequencing), and the system can be crowd-sourced and expanded. It also has a strong educational and outreach potential. Our sampling system has been successfully tested in a lagoon, and has generated preliminary data.
What are the goals of the project?
My immediate goal is to deploy another sampling set in the summer or early fall, when beach water is warmer, to compare with our spring set. Plastic samples are placed in stainless steel cages (similar to lobster traps) and deployed in beach water. Samples are collected weekly for a month. DNA is extracted from the plastic samples and analyzed through next generation sequencing, which describes the total genetic makeup of the population attaching to plastic. I also take swabs from the plastic and use microbiological methods to characterize them. If done early enough I may be able to add the results to my conference presentation in August. Long-term goals include publication of the data and expanding to more beaches, ideally through crowdsourcing.
Budget
I started this project with two internal grants from my institution, which allowed me to complete two sampling runs, one for validation in 2015, and another in the spring of 2016. The funds via Experiment would allow me to complete another sampling set in the summer or early fall of 2016, when ocean temperatures are warmer, which would constitute a great comparison. I am currently applying to other external grants and awaiting the analysis of the latest samples. Those results will be presented at the 2016 ISME conference in August. Having 2 comparable datasets in the same beach location would enhance our odds to get external funding.
Update May 23, 2017. We just had one round of scanning electron microscopy of plastic samples with amazing results. As we move forward with this project, we realize the impact of microscopy to actually "see" and document both the state of plastic and the microbes attached to it.
Endorsed by
 Project Timeline
Project Timeline
Jul 07, 2016
Project Launched
Jun 04, 2017
Present at the ASM Microbe conference
Aug 31, 2017
Complete NGS sequencing of 2nd complete run
Meet the Team
Ana Maria Barral
My scientific journey started with the study of biochemistry, as I was interested in the internal chemical workings of living organisms. I got my bachelor's at the University of Havana, Cuba; and went on to work on cancer and tumor cell biology. Ph.D. studies took me to Sweden, and postdoc work to Southern California (La Jolla Institute for Allergy & Immunology), where I found my new home. After a brief stint at a biotech company it was clear to me that my heart was in teaching, and in 2012 became Assistant professor at National University, a California private non-profit specialized in accelerated learning. Over time I realized that I missed bench research, and also that coursework based on research is the best way to teach science. In the past years have been involved in a number of initiatives bringing authentic research to undergraduate classrooms, such as the Genomics Education Partnership, GCAT-SEEK, and the Small World Initiative, and learned the power and possibilities of next generation DNA sequencing and metagenomics. The ocean has always been a passion and interest of mine, so the idea to follow-up Emelia's studies exploring the microbes on microplastics on coastal plastic was extremely appealing to me. This project has the potential to become a rich citizen science and educational project, providing information useful for scientists, policy makers, and the general public.
Emelia DeForce
Emelia currently resides in Oceanside, CA and works at MO BIO Laboratories as a Research Scientist in R&D. She received her master’s and PhD degrees at University of Massachusetts Boston studying both medically important microorganisms as well as microbes that live in soil. During her postdoctoral work, she was investigating microbes that live in hydrothermal vent ecosystems and on the surface of plants. Because microorganisms are literally found everywhere, she has traveled to very remote places to sample, and is thankful for every opportunity to be on the ocean. Emelia participated in Plastics at SEA: North Atlantic and North Pacific Expeditions where she worked to better understand the role that microbes play in plastic pollution in the ocean.
Wendy Ochoa
Dr. Wendy Ochoa is an esteemed professional and creative mind. She is able to unleash from the ordinary and tap into her remarkable ability to harmonize creation and science. She holds a PhD in biochemistry and owns CADE collective, an innovative marketing consulting company. She has over fifteen (15) years in academic research, employment in the Biotech industry and she is an adjunct professor in biology and chemistry. She is passionate about science, education and design. She is committed to applying her knowledge and talents in way that improves our world.
Rachel Simmons
I earned my PhD in Genetics at UC Davis, and a BS and MS in Biology with a specialization in Ecology, Behavior and Evolution at UC San Diego. Throughout my career I have been interested in biotechnology and ecology, and how to use biotech tools to better understand natural phenomena. I've worked at biotechnology firms Axiom Biotechnologies (now Sequenom), HK Pharmaceuticals and Scidera Inc. from the bottom up, starting as a summer laboratory assistant in tissue culture and finally a paid analysis consultant for dog pedigree tests. In my research I have used genetic tools to look at ecological and evolutionary patterns of diverse species, including slipper snails in Mission Bay, gibbons in Southeast Asia and several subspecies of rainbow trout in California. My current research and collaborations use statistical analyses in education assessment and genetic tools to estimate environmental microbial diversity in a wide array of settings, including the effects of native plant roots on microbial soil communities, differences in microbial community composition on ocean plastics, and testing for differences in microbial species on skin samples of whales. I welcome student participation in these projects or the opportunity to work with students on related projects of their own.
Additional Information
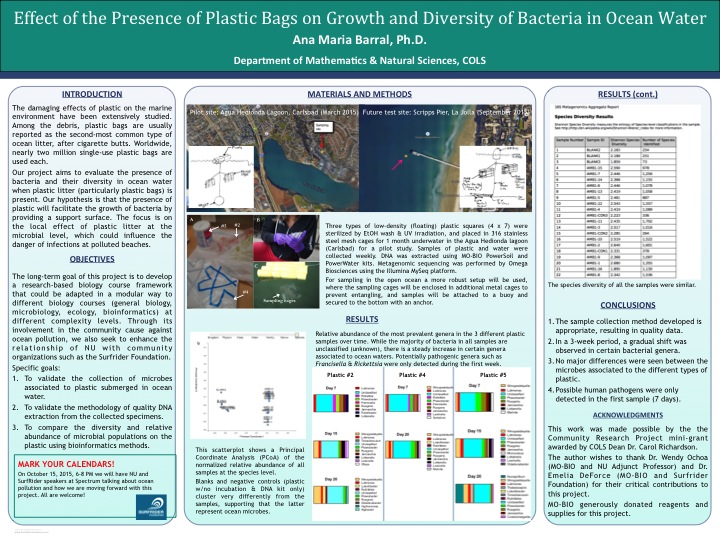
Here is a poster that I presented at my university's research symposium last year about the validation set:
 Here is a picture of the buoy anchored at Doheny State Beach, Dana Point, CA:
Here is a picture of the buoy anchored at Doheny State Beach, Dana Point, CA:

Here is a video taken during the spring deployment:
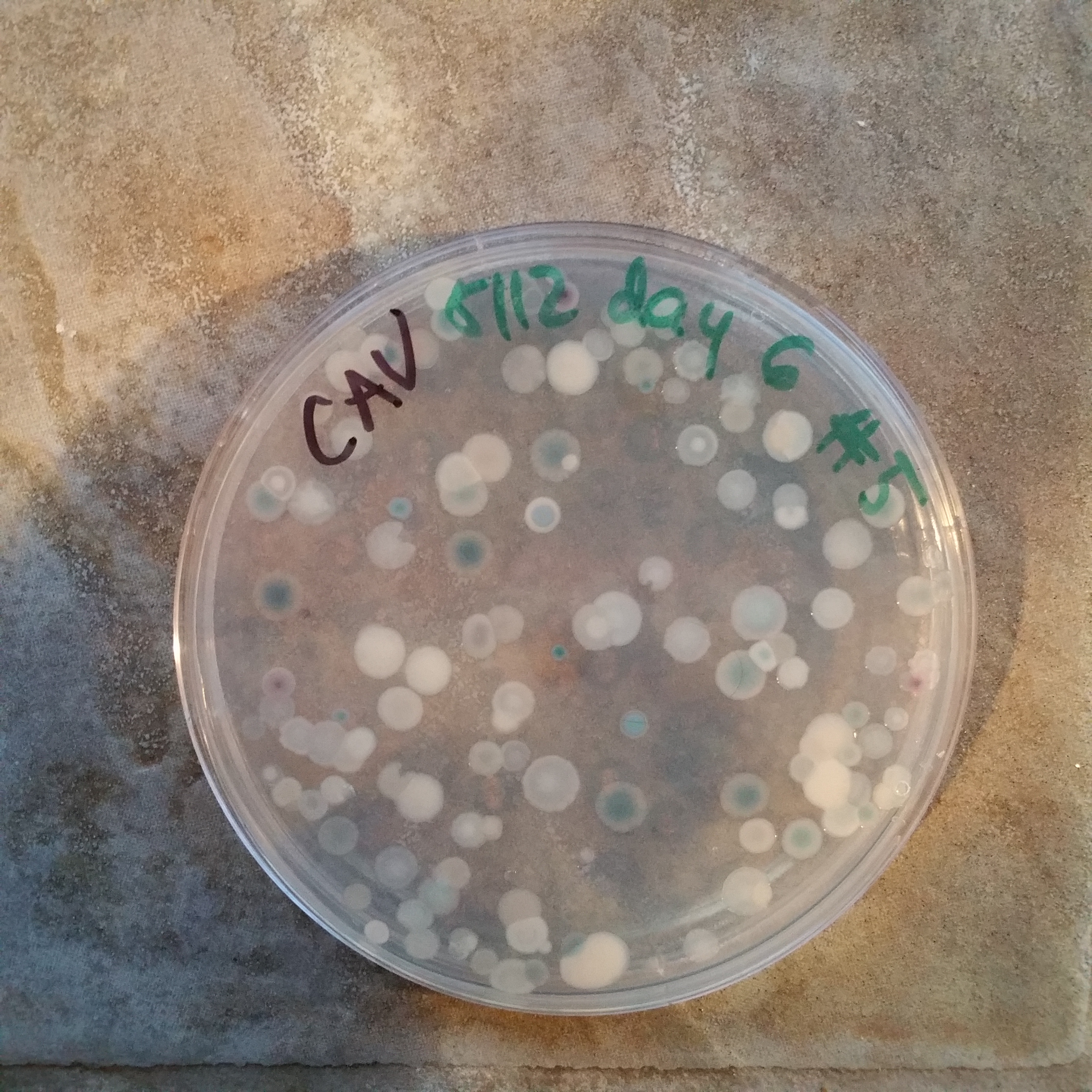
And a picture of the microbes growing on culture media. This was a 28-day sample of plastic #2 (high density polyethylene, found in grocery bags and other containers) on ChromAgar Vibrio, a selective-differential medium for Vibrios.
Project Backers
- 33Backers
- 101%Funded
- $2,020Total Donations
- $61.21Average Donation





